
Welcome to the new year with Genesis.
First of all, our thoughts go out to those affected by the bushfires here in Australia. Our deepest gratitude goes out to the incredible people fighting the fires, and to those providing relief to victims and wildlife.
This year, Genesis will continue to bring new clinical trial opportunities to our local community. We are looking forward to partnering with new investigators, expanding our trial site services, and creating more educational content.
Here’s to a great year for all.
This is a clinical trial opportunity for people living with early onset sciatic pain – pain that radiates into the leg as a result of disc herniation (bulging / slipped disc).
The study involves the injection of an investigational medicine into the spine that may offer longer lasting relief from sciatic pain than currently available treatments.
You may be eligible to join this study if you have been experiencing sciatic pain in one leg for between 1 to 6 months and you are aged between 18 and 65 years.
Contact us for more information.
We are conducting a clinical trial for chronic back and/or leg pain patients who are eligible to trial a commercially available high frequency spinal cord stimulator (SCS) device for their pain.
Patients who experience a successful trial of the SCS device and receive an implant will have the opportunity to trial advanced high frequency stimulation programs (“ultra-high frequency”), which may offer better pain control than standard high-frequency programming.
Playfully, yet appropriately, named ‘Subwave’, this trial of new SCS waveform programming began enrolling patients in 2018 and now has 12 implanted participants.
The results so far look promising and have recently been presented by Dr Russo at the 2020 North American Neuromodulation Society annual meeting in Las Vegas.
Contact us for more information.

Our first investigator-led network study is underway, with 5 participants already enrolled! The investigators of this study are some of Australia’s greatest pain specialists.
This is a study for chronic back pain patients who have not had success with or are no longer getting enough pain relief with spinal cord stimulation therapy. Eligible patients are given the option to trial low-dose intrathecal drug pump therapy as part of routine pain management. The drug pump is a commercially available implanted device that delivers medication to the spinal canal to treat chronic pain.

It was a great privilege for Genesis to be the first site to conduct a clinical trial for a new spinal cord stimulator device that delivers ultra-low frequency stimulation to treat chronic back pain.
We enrolled 15 participants onto the study, many of whom experienced life-changing improvements during their trial of the new device.

Last year, we successfully enrolled 100 healthy participants onto a clinical trial of an investigational blood pressure monitoring device.
The purpose of the new device is to improve the ease of use and accuracy of blood pressure monitoring at home. The data that was collected will be used to improve the software operating the device in order to provide accurate blood pressure readings.

Last December, our clinical trial coordinator, Michael, and investigator, Dr Uli Liedvogel, jetted off to San Diego, California, to attend an investigators meeting for a new osteoarthritis of the knee study that will be kicking off this year.
Genesis is one of five Australian sites that will be involved in the study. Research professionals from 46 U.S. sites also attended the meeting.
If you would like to know more about this upcoming study for osteoarthritis of the knee – please contact us!

Our clinical trial coordinators, Ashlie and Katie, were very fortunate to attend an investigators meeting with GlaxoSmithKline (GSK) at the Kuala Lumpur Convention Centre in December 2019.
During the two-day conference, Ashlie and Katie learnt about a new rheumatoid arthritis study and met various other clinical research professionals. The team are excited to bring the potential new therapy option to our local community in Newcastle in 2020.
If you would like to know more about this upcoming study for rheumatoid arthritis, please contact us!

Last year, we celebrated our Medical Director and Primary Investigator Dr Marc Russo’s 20th anniversary of opening his pain clinic in Newcastle.
The pain clinic also celebrated their rebranding to Hunter Pain Specialists (formerly Hunter Pain Clinic).

What a way to start the year! During our second week back, we spent a day looking forward for the business and then down the aisle of a bowling alley.
We kicked off the morning with a 2020 business planning session, then headed out for lunch and an afternoon of team building fun at Dullboy’s Social Co. We enjoyed some friendly rivalry, playing two games of tenpin bowling. There were strikes and spares all around.

Over the last 5 years, we have reached over 2.5 million people through social media! Through our Facebook page, we have connected people with local clinical trial opportunities.
Since establishing our Facebook page, we have expanded our site services, and now regularly provide participant recruitment services to other private clinical trial sites, major public hospitals, contract research organizations, and sponsors within Australia and New Zealand.

We are proud to announce that our GCP program that was internally developed and made live in 2019 has had over 50 users. Genesis offers a contemporary, accessible online resource that is not only free to undertake but meets the minimum criteria for ICH-GCP investigator site personnel training identified by TransCelerate BioPharma Inc, as necessary to enable mutual recognition of GCP training among trial sponsors.
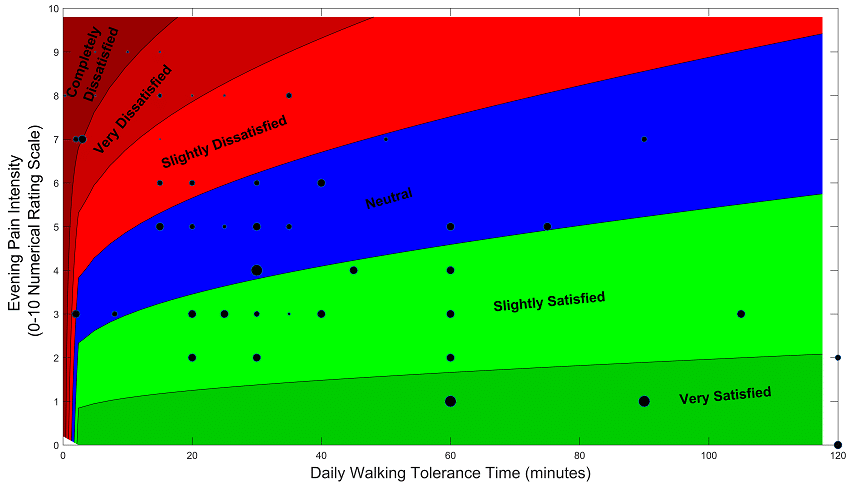
Congratulations to Dr Russo, Dr Verrills and co-authors on their publication on predicting patient satisfaction with spinal cord stimulation (SCS). In this paper, neuro-modulation researchers performed complex analysis of data from a clinical trial of SCS programs for chronic back pain patients.
The analysis showed that a combination of evening pain score and walking tolerance time may be best able to predict a patient’s satisfaction with SCS therapy.
The specific data analysed here showed that patients are likely to be satisfied if their pain score is low, or, if they can tolerate walking for longer periods of time, despite having a higher pain score.
View our current studies or register your interest for future trials: