

Neuromodulation represents an advanced pain management therapy for patients with chronic, refractory pain. The term “neuromodulation” refers to the therapeutic administration of electrical stimulation (or chemical agents) directly to the nervous system (spinal cord, brain or peripheral nerves) in order to modulate nerve cell activity. The aim is to interrupt pain signalling at the point of stimulation, thus stopping pain signals from reaching the brain.
The first sophisticated application of neuromodulation dates back to the early 1960’s, when deep brain stimulation (DBS) was first used to treat chronic, intractable pain. DBS involves the surgical implantation of electrodes within the brain that are wired to a pacemaker type device that is implanted under the skin on the chest. The device delivers electrical impulses to the target brain areas in order to mask abnormal activity. In the present day, DBS is primarily used in the treatment of neurological movement disorders, such as Parkinson’s disease, essential tremor and dystonia.
The application of spinal cord stimulation (SCS) commenced shortly after DBS, in 1967, and it is now the most established neurostimulation modality for the treatment of chronic, refractory pain, with a well-documented efficacy and safety profile. Traditional SCS produces tingling or “pins and needles” sensations, known as paraesthesia, to mask the pain.
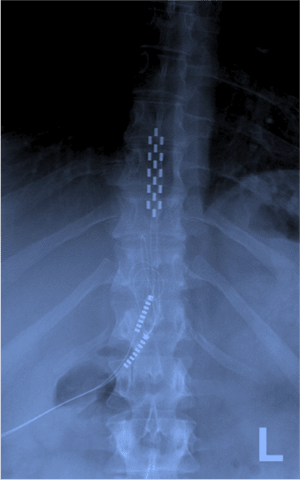
The two most common indications for SCS are failed back surgery syndrome (ongoing pain post back surgery) and complex regional pain syndrome (CRPS). The effectiveness of SCS is most well demonstrated in lower back and leg pain, although advancements in the technology are providing effective pain relief in a wider range of pain distributions.
SCS involves the implantation of leads into the epidural space that are connected to an implanted pulse generator (IPG), which is generally placed under the skin on the lower backside. The position of the stimulating leads along the spinal cord depends on the area of pain. For SCS systems that mask the pain by producing paraesthesia, the implanting physician will conduct “paraesthesia-mapping”. This requires the patient to communicate with the physician during lead placement in order to locate the position along the spinal cord which best provides coverage of their area of pain.
Before permanent implantation of the device, patients undergo a trial. Trial leads are implanted and connected to an external device. The trial usually lasts about a week, during which the patient completes a daily assessment of the SCS therapy. The trial is successful if the patient experiences a ≥50% decrease in pain, or significantly benefits in terms of disability and/or functionality. Many SCS patients are also able to reduce their analgesic medication intake.
Patients will need to attend a series of programming sessions with a technician from the SCS device company. During these sessions, stimulation parameters are tweaked and different stimulation programs are set for the patient to test or switch between if necessary. With regular programming, the relief gained from SCS can be optimised to suit the individual’s needs.
Nerves outside of the spinal cord can also be stimulated in order to treat chronic, refractory pain of the limbs. This is known as peripheral nerve stimulation (PNS). PNS is also used to treat conditions such as paraplegia and epilepsy. The target nerve is directly stimulated by implanting and anchoring an electrode on, near or around the nerve. Or, in the case of peripheral nerve field stimulation (PNFS), electrodes are placed under the skin within the site of most pain to stimulate the surrounding nerves.
Many patients will achieve significant benefits from traditional SCS systems. For patients who do not respond to traditional SCS, have a more complex pain distribution, or find paraesthesia uncomfortable, pain relief may be achieved with alternative, more recently developed SCS technologies.
High frequency SCS systems deliver significantly higher electrical stimulation – up to 10 kHz, compared to traditional stimulation which is delivered at around 50 Hz. This therapy does not produce paraesthesia, which is great for patients who find paraesthesia uncomfortable. Furthermore, the process of paraesthesia-mapping is not required during surgery, meaning shorter operating times. Both high frequency and traditional SCS are effective for leg pain, however, studies have shown high frequency SCS to be more effective in a greater percentage of patients with predominant back pain.
In these systems, high frequency stimulation is delivered in closely-spaced “bursts”, as opposed to traditional SCS in which electrical stimulation is delivered at a consistent, low frequency (“tonic” stimulation). This mode of SCS is also paraesthesia-free.
Dorsal root ganglia (DRG) are bundles of sensory nerve cell bodies that are located at the rear of the spinal vertebrae, next to the dorsal nerve roots which emerge from the spine. DRG regulate the entry of sensory information from the peripheral nervous system to the central nervous system. By targeting the DRG, pain signals originating from the limbs are interrupted before they enter the spinal cord. Studies suggest that this therapy is more effective for CRPS than traditional SCS systems.
Conventional SCS systems are pre-programmed to deliver a fixed level of stimulation. This is known as an open-loop system. In order to optimise the therapy in an open-loop system, the stimulation settings are manually adjusted by the device technician during programming sessions; Patients may be given a single fixed stimulation program, or several fixed stimulation programs which they may switch between depending on their needs and stimulation sensation. More recently, closed-loop SCS systems have been trialed. A closed-loop system is an adaptive system, meaning that the level of stimulation is automatically adjusted depending on nerve activity, body position etc. With a closed-loop SCS system, uncomfortable stimulation sensations (i.e. due to changes in body position) may be avoided, while the effectiveness of the therapy is maintained.
These are miniaturised SCS systems that do not contain an implanted battery. Instead, the entire implanted system consists of electrodes with a built-in receiver. The implantable device may be inserted into the spine, at the DRG or along a peripheral nerve via needle. The implant procedure is minimally invasive and can be performed without sedation. The battery and transmitter are contained in an external, wearable device. The implanted system is activated (pulse generated) when the external device is placed on the skin within a certain distance of the internal receiver. This system can be used as often as needed, as long as the external device is worn. This is in contrast to fully implantable systems, which may be constantly switched on and regularly charged via a remote device.
High Density (HD) SCS describes a new method of SCS programming that increases the dose of energy delivered to the spinal cord at a frequency higher than conventional SCS yet below high frequency SCS. HD programming is capable of providing reduced paraesthesia or paraesthesia-free pain relief. This is known as “subthreshold” or “subperception” stimulation, and it can be achieved within a conventional SCS system. Studies have shown that HD programming may be capable of restoring therapy in patients with conventional SCS systems who have lost effective pain relief. Further studies are necessary in order to increase our understanding of and identify the optimal programming parameters.
Genesis Research Services conducts clinical trials for numerous neuromodulation devices for various chronic pain conditions. If you would like to be informed about upcoming neuromodulation clinical trials, click here to view currently enrolling trials or register your interest. We will contact you to discuss new trials for which you may be eligible to participate in.
View our current studies or register your interest for future trials: